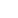Aseptic Isolator
This aseptic sterile isolator adopts the physical barrier method to provide isolation protection for the key operation process of sterile drugs, so as to minimize the risk of external environmental pollution of inspection products during operation and protect operators.
It provides a smooth, standardized and effective control process for the aseptic operation process, reduces the background environmental requirements of the aseptic clean room, simplifies the personnel dressing process, and reduces the operation cost.
Product Features:
1. Intelligent control system
2. Experimental operation area
3. VHP sterilization
4. Automatic chamber leak detection test
5. Integrated design
6. Internal bacteria collector
This aseptic isolator is designed in accordance with the related requirements of GMP, FDA, USP/EP. It’s with electrical record and electrical signature.
It’s equipped with two interlocked inflatable seal doors so as to make it nearly zero leak in the production.
The wind speed, pressure, temperature, relative humidity and hydrogen peroxide concentration in the chamber can be monitored in real time. Hydrogen peroxide concentration monitoring requires optional concentration sensors, it’s not a standard configuration.
The device supports real-time printing and storage of data.
This device can be operated both automatically and manually.
Power supply: AC380V 50HZ
Maximum power: 2500 Watts
Control system: NetSCADA system
Clean class: GMP Class A dynamic
Noise: < 65dB(A)
Lightness: >500Lux
Compressed air source: 0.5MPa ~ 0.7 MPa